
Trusted by 5000+ Manufacturers
Medical Software Compliance in Europe
Euverify helps medical software companies complete all mandatory steps to legally market Class I devices in the UK and EU from MHRA registration to EUDAMED/HPRA notifications and Authorised Representation.


What is Euverify and How Does It Work?

Core Features We Provide
Full UK Registration for Class I Medical Software
We handle your full MHRA registration and act as your UK Responsible Person so your Class I medical software can be legally sold in the UK. Our team ensures your documentation, labelling, and regulatory details meet all UKCA requirements from day one.


EU Market Access via EUDAMED
& HPRA
Euverify completes your EUDAMED Actor registration and HPRA device notification, unlocking legal access to the entire EU market. From SRN assignment to device listing, we manage every mandatory step required under EU MDR.
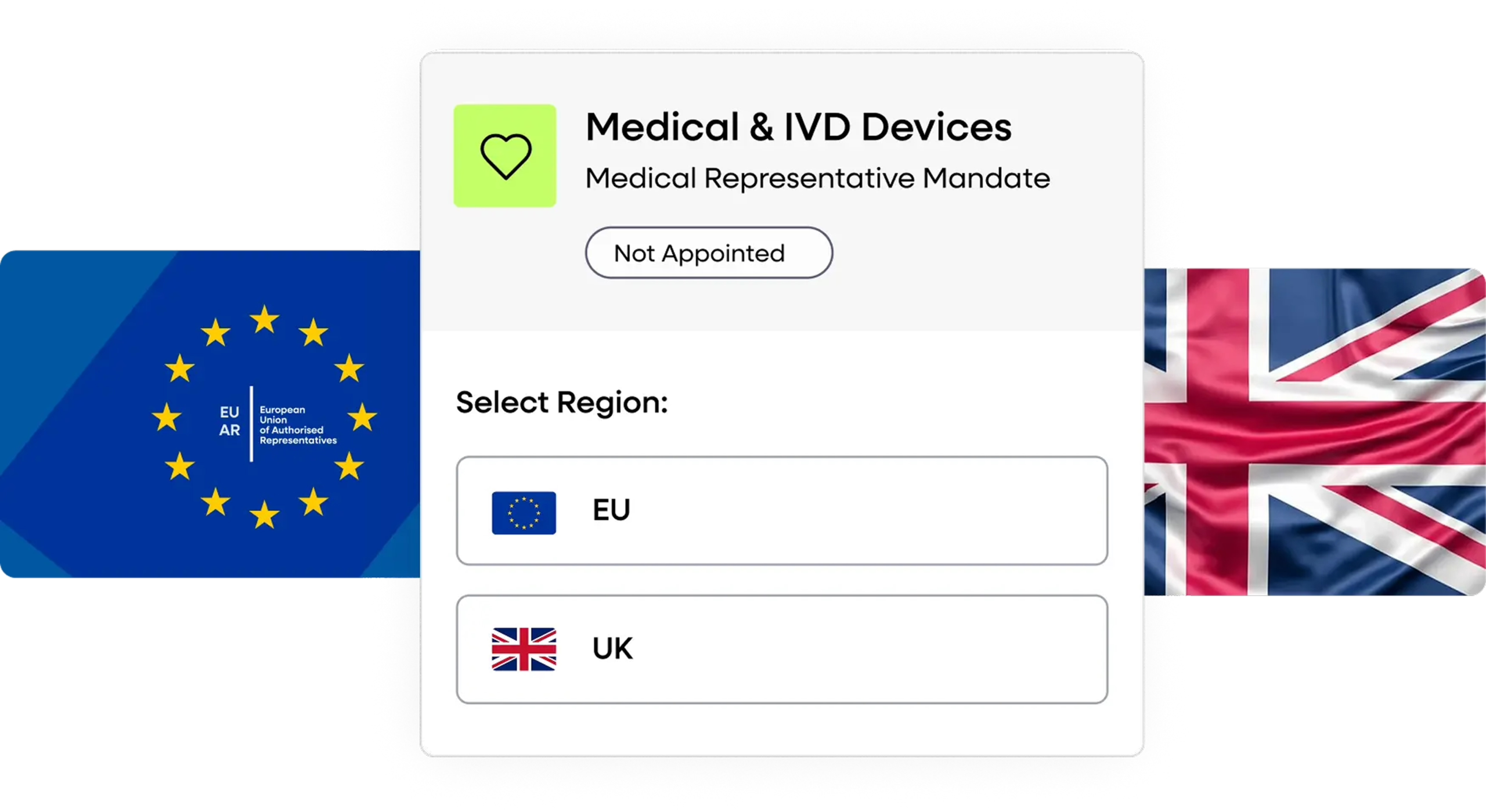
EU & UK Authorised Representative Services
If you don’t have a local presence, we act as your official EU Authorised Representative and UK Responsible Person. We handle all regulatory communication, document validation, and market surveillance inquiries on your behalf.



Technical Documentation Review for Software Devices
Our specialists review your entire technical file to ensure it aligns with MDR and UKCA requirements, including risk management, cybersecurity, usability, and clinical evaluation. We highlight gaps and guide you on how to complete or strengthen your documentation before registration.
GDPR Representative for Medical Software
If your software processes personal or health data in the EU or UK, we serve as your official GDPR Representative under Article 27. We manage data-protection inquiries, support DPIA readiness, and ensure your privacy policies meet regulatory expectations.


Ongoing Compliance Monitoring & Authority Support
After registration, we continue to support your regulatory needs by monitoring updates, reviewing changes, and responding to authority enquiries for you. Euverify keeps your documentation audit-ready and supports post-market compliance whenever required.
Support for Higher-Class Devices with Existing Certification
If you already hold a Notified Body certificate for Class IIa, IIb, or III software, we can act as your EU Authorised Representative and UK Responsible Person. We assist with registrations, documentation hosting, and ongoing regulatory communication for higher-class devices.


EU & UKCA Declaration of Conformity for Medical Software
We help you prepare fully compliant EU and UKCA Declarations for your Class I medical software. All MDR/UKCA references, applicable standards, risk details, and device information are accurately included. Our platform structures the document to meet regulatory expectations, helping you launch in the EU and UK with confidence.
Key Compliance Steps
Classify Your Software
Confirm it’s Class I under MDR/UKCA.
Prepare Technical Documentation
Risk file, cybersecurity, clinical evaluation.
Appoint EU & UK Representation
EU AR + UK Responsible Person (if no local address).
Complete Registrations
MHRA (UK), EUDAMED + HPRA (EU).
Create Declarations of Conformity
EU DoC & UKCA DoC.
Maintain Post-Market Compliance
Monitor updates, safety, and authority queries.
Which Types of Medical Software Need Registration?
Digital health apps and medical software used for diagnosis, monitoring, or treatment are regulated as medical devices in the UK and EU. Here’s a quick overview of how they fit into Classes I, IIa, IIb, and III.
Common Class I Medical Software
- Wellness & lifestyle tracking apps
- Symptom logging tools
- General health information apps
- Non-diagnostic monitoring apps
- Telehealth / video consultation platforms
- Appointment and patient management tools
- Medication reminder apps
- Rehabilitation & exercise guidance apps
- Software that records or stores medical data
- Basic image viewers (no diagnostic output)
See the full breakdown of Class I, Class IIa, Class IIb, and Class III medical software classifications and examples.
Why Choose Euverify?
Euverify gives medical software companies a fast, reliable, and fully guided path to UK and EU compliance. We handle every regulatory requirement in one place so you can launch confidently without delays or confusion.
What makes us different:
End-to-end registration for MHRA, EUDAMED, and HPRA
EU Authorised Representative & UK Responsible Person included
Technical documentation review tailored for medical software
GDPR Representative for EU & UK health data requirements
Fast onboarding become compliant in days, not months
Support for higher-class devices if you already have NB certification
Clear pricing with no hidden fees
Responsive compliance experts available when you need help
Pricing & Registration Enquiry
Get a personalised quote for your medical software registration in the UK and EU. Share a few details about your product, and our compliance team will respond with the exact pricing and next steps.
Request Your Pricing
A compliance specialist will review your details and send you a tailored quote within 24 hours.
Loved & Trusted by Businesses Worldwide
Trusted by 5000+ Brands
Frequently Asked Questions
If your software monitors, tracks, records, or influences any aspect of a person’s health, it must be registered as a medical device. Class I software (non-diagnostic, non-critical) requires MHRA/EUDAMED/HPRA registration before it can be sold in the UK or EU.
Class I includes low-risk software such as symptom trackers, wellness apps, telehealth platforms, appointment apps, medical record organisers, or any software that stores health data without interpreting it.
Class I devices are low risk and can be self-certified.
Class IIa and above are medium–high risk and require a Notified Body audit and certification. Euverify supports these only if the manufacturer already holds NB approval.
Yes — if you already have Notified Body certification, Euverify can act as your EU Authorised Representative and UK Responsible Person and complete all registrations (HPRA/EUDAMED/MHRA).
MHRA registration is mandatory for placing Class I medical software on the UK market. Without MHRA approval, you cannot legally sell your software in the UK.
EUDAMED is the EU’s central medical device database. For Class I, manufacturers must complete Actor Registration to obtain an SRN. Euverify handles the entire registration process for you.
Ireland (HPRA) is required because the Authorised Representative must register the device in the EU through their competent authority. Euverify handles HPRA notifications on your behalf.
No. Class I software typically does not require UDI assignment for EUDAMED or UKCA. Registration can be completed without a UDI for SaMD.
If you don’t have a legal address in the EU, you must appoint an EU AR. This is mandatory for all non-EU manufacturers selling software in the EU.
If your company is based outside the UK, you must appoint a UK Responsible Person to legally sell your Class I device in the UK.
Your technical file must include:
- Device description
- Software architecture
- Cybersecurity documentation
- Risk management (ISO 14971)
- Usability (IEC 62366)
- Clinical evaluation
- PMS plan
- Labelling & IFU
Euverify reviews all required documents for compliance.
Yes. Euverify prepares both EU Declaration of Conformity and UKCA Declaration, ensuring all MDR/UKCA references, standards, and device details are correctly included.
Yes. We serve as your EU & UK GDPR Representative (Article 27) and assist with privacy compliance for apps processing personal or health data.
Most Class I software registrations can be completed within:
- UK: 2–5 days
- EU: 5–10 days (EUDAMED + HPRA)
Timelines depend on document readiness.
As your AR / UKRP, Euverify handles all communication from regulators, including document requests, safety questions, or market surveillance checks.
Yes. We ensure updated versions still comply and we update your registrations if necessary.
Most Class I software can justify clinical safety using literature or equivalence. Euverify reviews your clinical evidence and guides what is required.
No for Class I.
Yes for Class IIa/IIb/III (Notified Body required).
Euverify provides ongoing compliance monitoring, annual reviews, and updates when standards or regulations change.
Class I software is not routinely audited, but authorities can request documentation at any time. Euverify ensures everything is audit-ready.
Pricing depends on markets (UK, EU, or both) and number of devices. Use the enquiry form to get an exact quote in 24 hours.
Not usually. Minor updates do not require new registration. Significant functional changes must be reviewed for compliance impact.
Yes. UK-only apps need MHRA + UKRP only.
EU-only apps need EUDAMED + HPRA + EU AR.
Yes. Euverify specialises in SaMD (Software as a Medical Device) and handles all software-specific regulatory steps.
Yes. App store availability does not remove medical device requirements. Registration is mandatory if the software has a medical purpose.
Yes. We help maintain PMS documentation and respond to any safety or performance issues detected after launch.
Yes. If you’re unsure whether your software is Class I or higher, we provide a quick classification assessment.




